A Warming World
Once there was a world not so very different from our own.
There were occasional natural catastrophes, massive volcanic eruptions and, every once in a while, an asteroid would come barrelling out of the blue to do some damage.
But for the first billion years or so, it would’ve seemed like a paradise, the very personification of its name: The Goddess of Beauty.

Then things started to go horribly wrong.
The planet Venus, which once may have seemed like a heaven, turned into a kind of hell. The difference between the two can be a delicate balance, far more delicate than you might imagine.
Once things began to unravel, there was no way back.

Venus’s oceans are long gone. The surface is hotter than a broiling oven, hot enough to melt lead. Why? You might think it’s because Venus is 30% closer to the Sun than the Earth is, but that’s not the reason. Venus is completely covered by clouds of carbon dioxide and sulphuric acid; the latter keeps almost all the sunlight from reaching the surface. That ought to make Venus much colder than Earth.
So why is Venus scorching hot? It’s because the small amount of sunlight that trickles in through the clouds to reach the surface can’t get back out again. The flow of energy is blocked by the dense atmosphere of carbon dioxide. That carbon dioxide gas – or CO2 for short – acts like a smothering blanket to keep the heat in.
No one is burning coal or driving big petroleum guzzlers on Venus. Nature can destroy an environment without any help from intelligent life.
Venus is in the grip of a runaway greenhouse effect.
In 1982, the scientists and engineers of what was then the Soviet Union successfully landed Venera 13 on Venus. They managed to keep it refrigerated for over two hours, so it could photograph its surroundings and transmit the images back to Earth before the onboard electronics were fried.

Venus and Earth started out with about the same amount of carbon, but the two worlds were propelled along radically different paths, and carbon was the decisive element in both stories. On Venus, it’s almost all in the form of gas – carbon dioxide – in the atmosphere.
Most of the carbon on Earth has been stored for aeons in solid vaults of carbonate rock, like limestone and chalk. How? Volcanoes supply carbon dioxide to the atmosphere, and the oceans slowly absorb it. Working over the course of millions of years, microscopic algae harvest the carbon dioxide and turn it into tiny shells. They accumulate in thick deposits of chalk, or limestone. Other marine creatures take in carbon dioxide to build enormous coral reefs. And the oceans convert dissolved CO2 into limestone even without any help from life. As a result, only a trace amount is left as a gas in Earth’s atmosphere. Not even four-hundredths of one per cent.
Think of it – about four molecules out of every ten thousand. And yet, it makes the critical difference between a barren wasteland and a garden of life on Earth. With no CO2 at all, the Earth would be frozen. And with about twice as many, we’re still talking about only six molecules out of ten thousand, things would get uncomfortably hot and cause us some serious problems.
But never as hot as Venus; not even close. That planet lost its ocean to space billions of years ago. Without an ocean, it had no way to capture CO2 from the atmosphere and store it as a mineral. The CO2 from erupting volcanoes just continued to build up.
Today, that atmosphere is 90 times heavier than ours. Almost all of it is heat-trapping carbon dioxide. That’s why Venus is such a ferocious inferno – so hostile to life.
Earth, in stunning contrast to Venus, is alive. It breathes, but very slowly. A single breath takes a whole year.
When spring comes to the north, the forests inhale carbon dioxide from the air and grow, turning the land green. The amount of CO2 in the atmosphere goes down. When fall comes and the plants drop their leaves, they decay, exhaling the carbon dioxide back into the atmosphere. The same thing happens in the Southern Hemisphere at the opposite time of the year. But the Southern Hemisphere is mostly ocean. So it’s the forests of the north that control the annual changes in global CO2.
Earth has been breathing like this for tens of millions of years. But nobody noticed until 1958 when an oceanographer named Charles David Keeling devised a way to accurately measure the amount of carbon dioxide in the atmosphere. Keeling discovered the Earth’s exquisite respiration. But he also discovered something shocking – a rapid rise, unprecedented in human history, in the overall level of CO2, one that has continued ever since.
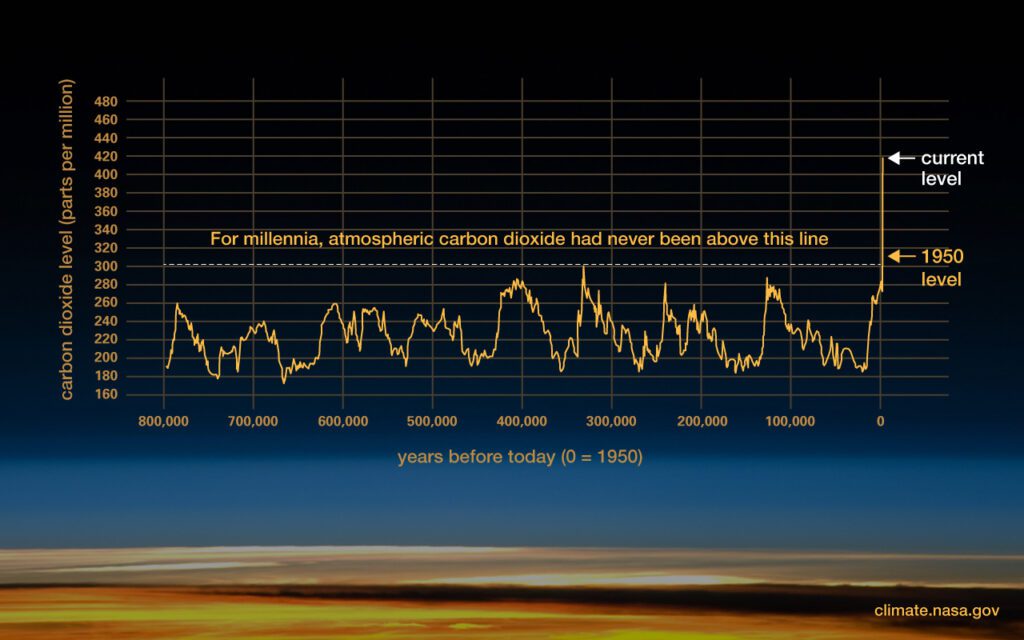
How can we be so sure? The evidence is written in water.
The Earth keeps a detailed diary written in the snows of yesteryear. Climate scientists have drilled ice cores from the depths of glaciers in Greenland and Antarctica. The ice layers have ancient air trapped inside them. We can read the unbroken record of Earth’s atmosphere that extends back over the last 800,000 years. In all that time, the amount of carbon dioxide in the air never rose above three-hundredths of one percent. That is, until the turn of the 20th century. And it’s been going up steadily and rapidly ever since. It’s now more than 40% higher than before the Industrial Revolution. By burning coal, oil and gas, our civilization is exhaling carbon dioxide much faster than Earth can absorb it. So CO2 is building up in the atmosphere. The planet is heating up.
Every warm object radiates a kind of light we can’t see with the naked eye—thermal infrared light. We all glow with invisible heat radiation, even in the dark.

Incoming light from the Sun hits the surface. The Earth absorbs much of that energy, which heats the planet up and makes the surface glow in infrared light. But the carbon dioxide in the atmosphere absorbs most of that outgoing heat radiation, sending much of it right back to the surface. This makes the planet even warmer.

If we didn’t have any carbon dioxide in our atmosphere, the Earth would just be a great big snowball, and we wouldn’t be here. So, a little greenhouse effect is a good thing. But a big one can destabilize the climate and wreck our way of life.
All right but how do we know that we’re the problem? Maybe the Earth itself is causing the rise in CO2. Maybe it has nothing to do with the coal and oil we burn. Maybe it’s those damn volcanoes. They’ve already doomed the planet Venus anyway.

Now, combine that with the output of all the other volcanic activity on the planet. Let’s take the largest scientific estimate – about 500 million tonnes of volcanic CO2 entering the atmosphere every year. Sounds like a lot, right? But that’s not even two percent of the 36 billion tonnes of CO2 that our civilization is cranking out every year. And, funny thing, the measured increase in CO2 in the atmosphere tallies with the known amount we’re dumping thereby burning coal, oil and gas. Volcanic CO2 has a distinct signature – it’s slightly heavier than the kind produced by burning fossil fuels. We can tell the difference between the two when we examine them at the atomic level. It’s clear that the increased CO2 in the air is not from volcanoes. What’s more, the observed warming is as much as predicted from the measured increase in carbon dioxide.
It’s a pretty tight case. Our fingerprints are all over this one.
How much is 36 billion tonnes of CO2 per year? If you compressed it into solid form, it would occupy about the same volume as Mount Kilimanjaro. And we’re adding that much CO2 to the air every year, relentlessly, year after year.

Unlucky for us, the main waste product of our civilization is not just any substance. It happens to be the chief climate-regulating gas of our global thermostat, year in, year out. Too bad CO2 is an invisible gas. Maybe if we could see it, if our eyes were sensitive to CO2 – and perhaps there are such beings in the cosmos – if we could see all that carbon dioxide, then we would overcome the denial and grasp the magnitude of our impact on the atmosphere.
But the evidence that the world is getting warmer is all around us. For starters, let’s just check the thermometers; Weather stations around the world have been keeping reliable temperature records since the 1880s, and NASA has used the data to compile a map tracking the average temperatures around the world through time.
As far back as 1896, Swedish scientist Svante Arrhenius calculated that doubling the amount of CO2 in the atmosphere would melt the Arctic ice. In the 1930s, the American physicist E.O. Hulburt, at the Naval Research Laboratory, confirmed that result. So far, it was still just theoretical. But then, the English engineer Guy Callendar assembled the evidence to show that both the CO2 and the average global temperature were actually increasing.
If we don’t change our ways, what will the planet be like in our children’s future? Based on scientific projections, if we just keep on doing business as usual, our kids are in for a rough ride:
- killer heat waves
- record droughts
- terminal tropical and highly infectious diseases in the far reaches of the globe
- mass extinction of species
- rising sea levels and sinking coastal cities
- mass death of coral reefs by ocean warming
- increase in the intensity of catastrophic storms
- runaway wildfires
We inherited a bountiful world made possible by a relatively stable climate. Agriculture and civilization flourished for thousands of years. And now, our carelessness and greed put all of that at risk.
Okay, so if scientists are so good at making these dire, long-term predictions about the climate, how come they’re so lousy at predicting the weather? Besides, this year, we are having a colder season in my country. For all scientists know, we could be in for global cooling.
Here’s the difference between weather and climate: Weather is what the atmosphere does in the short term – hour to hour, day to day. Weather is chaotic, which means that even a microscopic disturbance can lead to large-scale changes. That’s why those ten-day weather forecasts are useless. A butterfly flaps its wings in Kinshasa, and six weeks later, your outdoor wedding in Zanzibar is ruined.
Climate is the long-term average of the weather, over several years. It’s shaped by global forces that alter the energy balance in the atmosphere, such as changes in the Sun, the tilt of the Earth’s axis, the amount of sunlight the Earth reflects back to space and the concentration of greenhouse gases in the air. A change in any of them affects the climate in broadly predictable ways.
Climate has changed many times in the long history of the Earth but always in response to a global force. The strongest force driving climate change right now is the increasing CO2 from the burning of fossil fuels, which is trapping more heat from the Sun. All that additional energy has to go somewhere. Some of it warms the air. Most of it ends up in the oceans. All over the world, the oceans are getting warmer. It’s most obvious in the Arctic Ocean and the lands that surround it.

Ice is the brightest natural surface on Earth, and open ocean water is the darkest. Ice reflects incoming sunlight back into space. Water absorbs sunlight and gets warmer, which melts even more ice, which exposes still more ocean surface to absorb even more sunlight. This is what we call a positive feedback loop. It’s one of many natural mechanisms that magnify any warming caused by CO2 alone.
The Arctic Ocean is warming at an increasing rate. So it’s ice-free during more of the year. That leaves the shore more exposed to erosion from storms, which are also getting more powerful, another effect of climate change.

The northern reaches of Alaska, Siberia and Canada are mostly permafrost, ground that has been frozen year-round for millennia. It contains lots of organic matter, old leaves and roots from plants that grew thousands of years ago. Because the Arctic regions are warming faster than anywhere else on Earth, the permafrost is thawing and its contents are rotting, just like when you unplug the freezer. The thawing permafrost is releasing carbon dioxide and methane, an even more potent greenhouse gas, into the atmosphere. This is making things even warmer, another example of a positive feedback mechanism. The world’s permafrost stores enough carbon to more than double the CO2 in the atmosphere. At the rate we’re going, global warming could release most of it before the end of the century. We might be tipping the climate past a point of no return into an unpredictable slide.

No, it’s not the Sun. Scientists have been monitoring the Sun very closely for decades, and the solar energy output hasn’t changed. What’s more, the Earth is warming more at night than in the daytime, and more in winter than in summer. That’s exactly what we expect from greenhouse warming, but the opposite of what increased solar output would cause. It’s now clear beyond any reasonable doubt that we are changing the climate.
The Sun isn’t the problem. But it is the solution. In all its glory, the Sun pours immaculate, free energy down upon us; more than we will ever need; More solar energy falls on Earth in one hour than all the energy our civilization consumes in an entire year. The winds themselves are solar-powered because our star drives the winds and the waves. Unlike solar collectors, wind farms take up very little land, and none at all, if offshore, where the winds are strongest. If we could tap even one per cent of the available solar and wind power, we’d have enough energy to supply all our energy needs forever, and without adding any carbon to the atmosphere.
It’s not too late. There’s a future worth fighting for. How do I know? Every one of us comes from a long line of survivors. Our species is nothing if not adaptive. It was only because our ancestors learned to think long-term and act accordingly, that we’re here at all. We’ve had our backs to the wall before, and we came through to scale new heights. In fact, the most mythic human accomplishment of all came out of our darkest hour.

They gave up the ceaseless wandering, hunting and gathering that had been their way of life for a million years or so, to settle down and produce food. They found a way to harvest ten to a hundred times more solar energy than the environment naturally provided for their ancestors. People all over the world made the difficult transition from nomadic cultures to agricultural ones that used solar energy more efficiently. It gave rise to civilization. We stand on the shoulders of those who did the hard work that such a fundamental transformation required.
Now it’s our turn.
If life ever existed on Venus, it would have had no chance to avert the hellish destiny of that world. The runaway greenhouse effect was unstoppable.
Earth is our world. And that world is now. There are no scientific or technological obstacles to protecting our world and the precious life that it supports. It all depends on what we truly value and if we can summon the will to act.
 English
English French
French German
German








